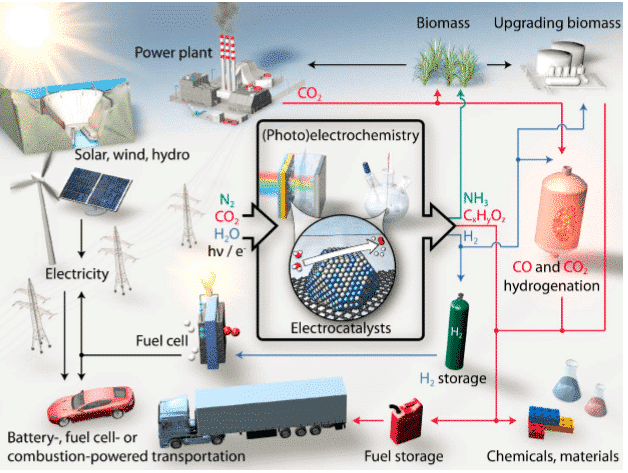
A tecnologia de reação eletrocatalítica é um dos métodos principais para conduzir essas vias de conversão de energia e purificação ambiental.
Nos últimos anos, com o desenvolvimento da sociedade e o progresso da humanidade, problemas energéticos e ambientais cada vez mais sérios se tornaram um problema mundial que precisa ser resolvido com urgência. As pessoas estão comprometidas com o uso eficaz de novas fontes de energia e métodos de purificação a longo prazo do meio ambiente. Os atuais métodos de pesquisa eficazes para promover a conversão de energia e a purificação ambiental envolvem muitas direções, como desenvolvimento de células de combustível, produção de hidrogênio, recursos de CO2, conversão catalítica orgânica de gases de escape. Os métodos de teste eletroquímico como um guia teórico fornecem um meio racional de interpretação para o desenvolvimento do desempenho do eletrocatalisador. Este artigo resume os métodos de teste eletroquímicos comumente usados em várias reações eletroquímicas.
Figura 1 Processo eletrocatalítico de conversão de energia sustentável
1. voltametria cíclica
A voltametria cíclica (CV) é o método de pesquisa mais comumente usado para avaliar sistemas eletroquímicos desconhecidos. É obtido principalmente controlando o potencial do eletrodo em diferentes taxas e digitalizando uma ou mais vezes com uma forma de onda triangular ao longo do tempo. Curva de potencial de corrente (IE). Diferentes reações de redução e oxidação podem ocorrer alternadamente nos eletrodos em diferentes faixas de potencial. A reversibilidade da reação do eletrodo pode ser julgada de acordo com a forma da curva; os picos de adsorção e dessorção dos reagentes podem ser usados para avaliar o eletrocatalisador de acordo com a faixa de potencial específica. A área cataliticamente ativa também pode ser usada para obter informações úteis sobre reações complexas de eletrodos.
Figura 1.1 Curva de resposta potencial atual de varredura
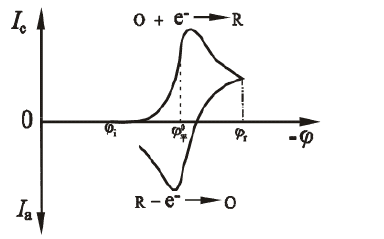
Como mostrado na Fig. 1.1, o potencial da primeira metade é varrido em direção ao cátodo e a substância eletroativa é reduzida no eletrodo para gerar uma onda de redução. Quando o potencial da segunda metade é digitalizado em direção ao ânodo, o produto de redução é novamente oxidado no eletrodo para gerar uma onda de oxidação. Dois parâmetros úteis da curva de voltametria cíclica iE são a razão de corrente de pico ipa / ipc e a diferença de potencial de pico Epa-Epc. Para a onda Nernst do produto estável, a taxa de corrente de pico ipa / ipc = 1, independente da velocidade de varredura, coeficiente de difusão e potencial de comutação. Quando a varredura de cátodo é parada, a corrente é degradada para 0 e, em seguida, reversa. A curva iE obtida é exatamente igual à curva do cátodo, mas é desenhada na direção oposta da coordenada I e da coordenada E. A razão ipa / ipc desvia de 1, indicando que o processo do eletrodo não é um processo de reação totalmente reversível que envolve cinética homogênea ou outras complicações. A altura do pico de reação e a área do pico podem ser usadas para estimar parâmetros do sistema, como a concentração de espécies eletroativas ou a constante de velocidade da reação homogênea acoplada. No entanto, a curva CV não é um método quantitativo ideal, e seu uso poderoso está mais em sua capacidade qualitativa de julgamento semi-quantitativo.
Voltametria 2.pulse
A voltametria de pulso é um método de medição eletroquímico baseado no comportamento dos eletrodos polarográficos. É usado para estudar o processo redox em vários meios, a adsorção de materiais de superfície em materiais catalisadores e o mecanismo de transferência de elétrons na superfície de eletrodos quimicamente modificados. A detecção é especialmente eficaz. A voltametria de pulso inclui voltametria de passo, voltametria de pulso convencional, voltametria de pulso diferencial e voltametria de onda quadrada, dependendo da maneira como a voltagem é varrida. Entre eles, a voltametria de passo é semelhante ao método de varredura potencial, e a resposta da maioria dos sistemas ao passo de volt-ampère de resolução mais alta (ΔE <5 mV) é muito semelhante aos resultados do experimento de varredura linear da mesma velocidade de varredura.
3. espectroscopia de impedância eletroquímica
A espectroscopia de impedância eletroquímica é aplicar um sinal elétrico perturbado ao sistema eletroquímico. Ao contrário do método de varredura linear, o sistema eletroquímico está longe do estado de equilíbrio e, em seguida, a resposta do sistema é observada, e as propriedades eletroquímicas do sistema são analisadas pelo sinal elétrico de resposta. A espectroscopia de impedância eletroquímica é frequentemente usada para analisar, avaliar a reação ORR em células a combustível PEM, caracterizar a perda de difusão na superfície do material catalisador, estimar a resistência ôhmica e as características da impedância de transferência de carga e capacitância de dupla camada para avaliar e otimizar o conjunto de eletrodo de membrana.
O espectro de impedância é geralmente desenhado na forma de um diagrama de Bode e um diagrama de Nyquist. No diagrama de Bode, a magnitude e a fase da impedância são plotadas em função da frequência; no diagrama de Nyquist, a parte imaginária da impedância é plotada em cada ponto de frequência em relação à parte real. O arco de alta frequência reflete a combinação da capacitância de camada dupla da camada de catalisador, a impedância efetiva de transferência de carga e a resistência ôhmica, que reflete a impedância produzida pela transferência de massa. Para um determinado sistema, as duas regiões às vezes não são bem definidas.
Figura 3.1 Espectro de impedância do sistema eletroquímico
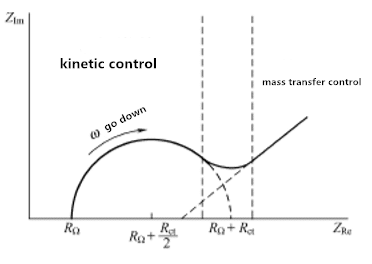
A Figura 3.1 mostra as características extremas do controle cinético e controle de transferência de massa. No entanto, para qualquer sistema, as duas regiões provavelmente não estão bem definidas. O fator determinante é a relação entre a resistência de transferência de carga e a impedância de transmissão. Se o sistema químico for lento em cinética, mostrará um Rct grande, que parece ter uma região de frequência muito limitada. Quando o sistema é dinâmico, a transferência de material sempre desempenha um papel de liderança e as áreas semicirculares são difíceis de definir.
4. cronoamperometria
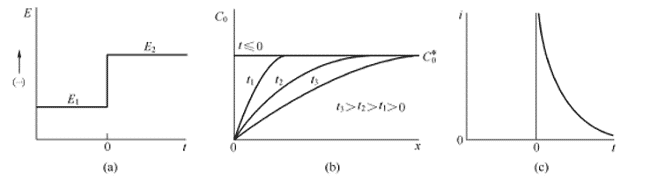
O método de cronoamperometria é um método de controle transitório que pode ser usado para avaliar a adsorção e difusão da superfície do catalisador. A curva de cronoamperometria é obtida aplicando uma etapa potencial ao sistema eletroquímico para medir a alteração do sinal de resposta atual ao longo do tempo. Quando um passo em potencial é dado, a forma de onda básica é mostrada na Figura 4.1 (a), e a superfície do eletrodo sólido é analisada com um material eletroativo. Após a aplicação do passo em potencial, as espécies eletroativas próximas à superfície do eletrodo são primeiro reduzidas a um radical ânion estável, que requer uma grande corrente, pois o processo ocorre imediatamente no instante do passo. A corrente que flui posteriormente é usada para manter as condições sob as quais o material ativo da superfície do eletrodo é completamente reduzido. A redução inicial causa um gradiente de concentração (ou seja, concentração) entre a superfície do eletrodo e a solução a granel, e o material ativo começa a se difundir continuamente em direção à superfície e difundir no eletrodo. O material ativo na superfície é completamente reduzido imediatamente. O fluxo de difusão, ou seja, a corrente, é proporcional ao gradiente de concentração da superfície do eletrodo. No entanto, note-se que, à medida que a reação prossegue, o material ativo na solução a granel difunde-se continuamente em direção à superfície do eletrodo, fazendo com que a região do gradiente de concentração se estenda gradualmente em direção à solução a granel e o gradiente de concentração da superfície do eletrodo sólido gradualmente fica menor (esgotado) e a corrente muda gradualmente. pequeno. A distribuição da concentração e a corrente versus o tempo são mostradas na Figura 4.1 (b) e na Figura 4.1 (c).
Figura 4.1 (a) Forma de onda experimental de etapa, reagente O não reage no potencial E1, é reduzida em E2 na velocidade limite de difusão; (b) distribuição da concentração em momentos diferentes; (c) curva corrente versus tempo
5. tecnologia de eletrodo de disco rotativo
A tecnologia de eletrodo de disco rotativo (RDE) é muito útil no estudo da reação homogênea acoplada da superfície do catalisador, de modo que a reação eletroquímica na superfície do catalisador é realizada sob uma condição de estado relativamente estável. O RDE pode controlar substâncias com difusão mais lenta, como o gás que se difunde facilmente na solução, reduzindo a influência da camada de difusão na distribuição de densidade atual. Assim, é obtida uma densidade de corrente estável, que está em um estado estacionário aproximado, o que é benéfico para o processo de análise eletroquímica; O RDE pode controlar a velocidade na qual o eletrólito atinge a superfície do eletrodo, ajustando a velocidade de rotação e medir os parâmetros do processo de reação eletrocatalítica em diferentes velocidades de rotação. análise.
À medida que os seres humanos se tornam mais interessados no desenvolvimento de eletrocatalisadores avançados para conversão de energia limpa, além de enfatizar o uso de alguns métodos básicos para a caracterização de reações eletrocatalíticas, é necessário um exame mais aprofundado das etapas elementares de cada reação para determinar os envolvidos. intermediários, a superfície do intermediário e a energia de cada etapa da reação elementar. O estudo de métodos eletroquímicos ainda exige muitos detalhes sobre a interface eletrodo-eletrólito que não são conhecidos até o momento, como as barreiras cinéticas e de reação envolvidas nas principais etapas elementares da transferência de próton / elétron; perto de solventes, cátions e interfaces de reação. A descrição atômica do estado de nível molecular do ânion; e os métodos de aquisição de sinal em tempo real mais rápidos e eficientes em todo o processo de reação eletroquímica ainda estão na vanguarda das reações eletrocatalíticas. Em resumo, o estudo aprofundado dos métodos de caracterização eletroquímica fornece uma estratégia orientadora para o desenvolvimento de novos sistemas catalisadores de alta eficiência.